|
9/20/2023 0 Comments Isotopes of chlorineChlorine has 25 isotopes, 2 of them are stable and naturally occurring.Ĭhlorine is used in water purification, bleaches, acids and many other compounds such as chlorofluorocarbons (CFC). Humphrey David confirmed chlorine as an element in 1810. Chlorine was discovered by Karl Scheele in 1774. It reacts directly with many elements and compounds, strong oxidizing agent. The longest-lived radioactive isotope is 36 Cl (half-life of 301,000 years) all other isotopes having half-lives of less than 1 h. There are only two stable isotopes: 35 Cl and 37 Cl with respective proportions of 75.76 and 24.24 (Berglund and Wieser 2011 ). 
This element occurs widely in nature as sodium chloride in seawater. Chlorine has 24 isotopes with mass numbers ranging from 28 Cl to 51 Cl. 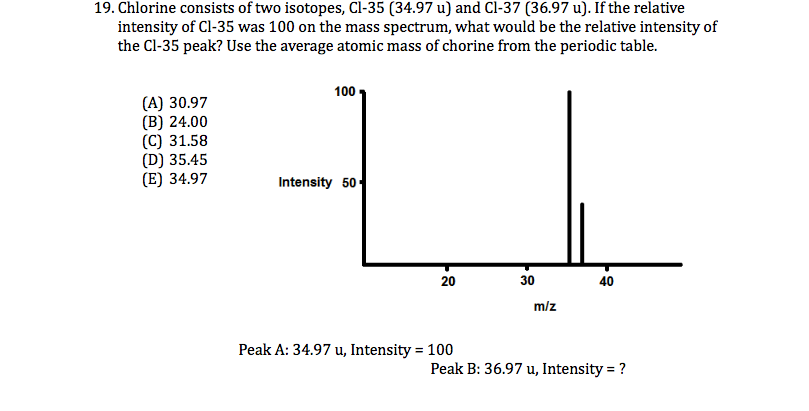
Please contact us via request a Chlorine-37 isotope quote to order Chlorine-37 isotope, to get Chlorine-37 price and to buy Chlorine-37 isotope.īack to list of enriched isotopes in alphabetical orderĬhlorine is a halogen element. CHLORINE-37 isotope is used for production of Sulphur-38 (38S isotope, S-38 isotope, Sulfur-38 isotope) radionuclide (radioisotope) for usage as a corrosion tracer (Industrial uses of radioisotopes and enriched isotopes) Ĭhlorine-37 isotope is available to order from in Chlorine-37 sodium chloride (NaCl) chemical form.Therefore the isotope of Chlorine with atomic mass 35 and 37 exists in the ratio of 3: 1. Step 5: calculating the ratio of the relative abundance of two isotopes: The ratio of Chlorine with atomic mass 35 to Chlorine 37 75: 25 3: 1. We can also write elements as EA where the E is the element symbol and the A is the atomic mass of. We can calculate, the relative abundance of Chlorine with an atomic mass of 37 100 x 100 75 25. The chemical properties of the different isotopes of an element are the same, but they might vary in how stable their nucleus is. The longest-lived radioactive isotope is 36 Cl (half-life of 301,000 years) all other isotopes having half-lives of less than 1 h. Isotopes of an element have the same number of protons (same Z), but a different number of neutrons (different N). 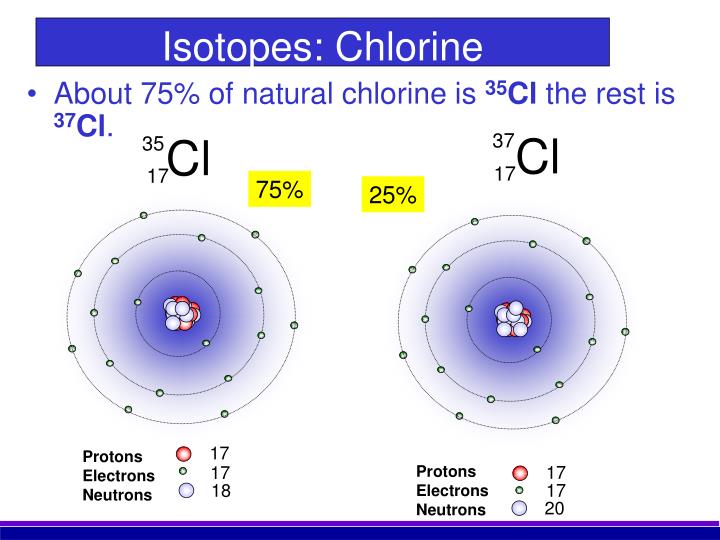
There are only two stable isotopes: 35 Cl and 37 Cl with respective proportions of 75.76 and 24.24 (Berglund and Wieser 2011). Please contact us via request a Chlorine-35 isotope quote to order Chlorine-35 isotope, to get Chlorine-35 price and to buy Chlorine-35 isotope.Ĭhlorine-37 isotope ( Cl-37 isotope, 37Cl isotope) Chlorine has 24 isotopes with mass numbers ranging from 28 Cl to 51 Cl. biological research and life science Ĭhlorine-35 isotope is available to order from in Chlorine-35 sodium chloride (NaCl) chemical form. In contrast, Cl has 2 common isotopes, 35Cl and 37Cl.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
